
Our Story
A brilliant biochemist and veteran of the biotechnology industry, Dr. Ram Bhatt left his homeland at a young age to pursue an education with hopes of one day solving the problem of the blood brain barrier. 35 years of scientific and executive experience in the biotechnology industry later, Dr. Bhatt started his own company to tackle the challenge in 2008. Just three years later, Dr. Bhatt and his team had proven the principle that a molecule directed to a target could enter and pass through the Blood Brain Barrier, unlocking a new frontier in the treatment of neurodegenerative disorders.
Our Mission
To design and develop innovative technologies capable of optimizing, cloning and mass production. We are committed to aiding in the fight against major diseases faced by humans all around the world today


Our Science
Our SMART Molecule Technology Platform (Specific Molecular Architecture for Recognition and Therapy) is a stripped down antibody mimic capable of identifying and eradicating any disease that leaves a target protein in the body.
Our Impact
Our current focus is on Parkinson’s and Alzheimer’s due to our desire to demonstrate our ability to penetrate the blood brain barrier. Proving our ability to do so now points at our ability to get through any tissue in the body, and bring Smart Molecules with low bulk and high target specificity to fight against virtually any major disease in the human body.

The Innovative California Biosciences International (ICBI) Team
Advisory Team

Dr. Robert Y. Ning, Ph.D., MBA
Dr. Ning has worked in the pharmaceutical industry for over 25 years, most recently as Vice President of Science and Business Development, and has published numerous papers on organic and process chemistry.

Dr. Anna Cartier, Ph.D.

Dr. Jayaraman A. Rao, MD

Jo Rosen
As the former President and CEO of R J Romero Financial Management Services, Inc., Ms. Rosen brings with her an abundance of business and financial management skills that have been very successful. Managing and overseeing the management of a multitude of clients, Ms. Rosen skillfully turned clients in crisis into clients of means. With her near three decades of experience as President and Executive Director of Parkinson’s Resource Organization, Ms. Rosen personally met the challenges of Parkinson’s with her late mother and her late husband. Her breadth of knowledge about the disease, living with the disease with the greatest quality of life, and the science that has been pervading the Parkinson’s world for these decades is equal to the ICBII dedication and determination to bring the reversal of Parkinson’s to this world.
Latest Press
ICBII Announces Approval of Its 7th Patent on Blood-Brain Barrier Permeable Technology, Moving Closer to Clinical Trials on its drugs for Alzheimer’s, Parkinson’s, and Other Neuro-Degenerative Diseases
2019 Market Report: Novel Technologies for Delivery of Proteins, Antibodies and Nucleic Acids, 2019-2030
ICBII announces approval of two US patents and filing of two divisional patent applications in Europe.
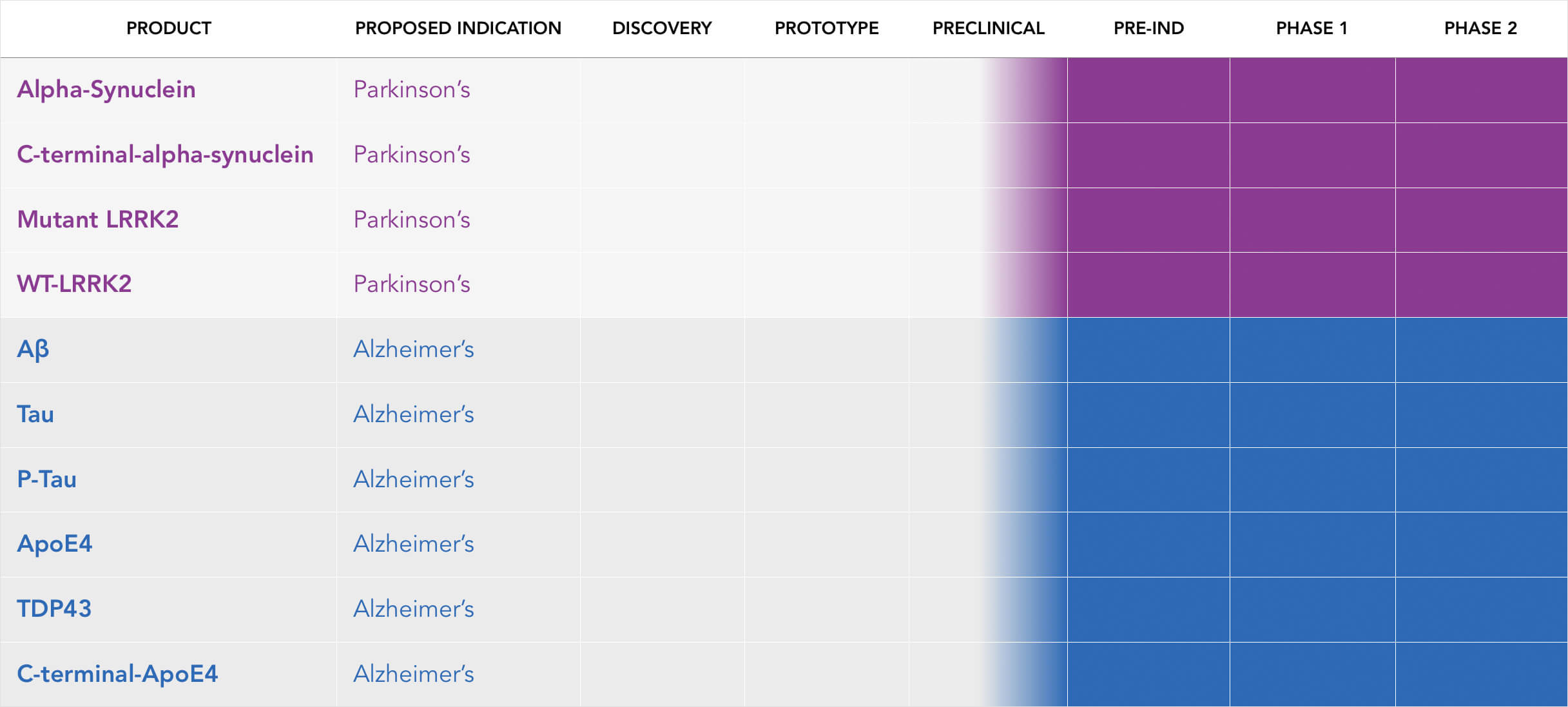
ICBI Neurodegenerative Pipeline
We are in preclinical studies for Parkinson’s disease and Alzheimer’s disease. As a platform technology, we intend to pursue studies where we believe our SMART molecules can address other debilitating diseases including brain cancer, colon cancer, breast cancer and prostate cancer.
IP Portfolio
Approved Patents
- Methods for using antibodies and analogs thereof: EP 2350655
- Blood-brain barrier permeable peptide compositions: EP 13000098.7
- Blood-brain barrier permeable peptide compositions: US 13/917,010
- Blood-brain barrier permeable peptide compositions: US 13/736,852
- Blood-brain barrier permeable peptide compositions: EP 13171872.8
Pending
- Methods for using antibodies and analogs thereof: US12/606,808
- Antibodies, analogs, and uses thereof: US 12/563,330
- Methods for using antibodies and analogs thereof: EP 09825245.5
- Methods for using antibodies and analogs thereof: EP: 16204570.2
- Blood-brain barrier permeable peptide compositions: EP 17187006.6
Publication
Molecular Imaging Insights into Neurodegeneration: Focus on α-Synuclein Radiotracers, Maliha Shah, John Seibyl, Anna Cartier, Ram Bhatt and Ana M. Catafau, J Nucl Med. 2014:55:1397-1400.

